Leonie Von Berlin et. al. Early fate bias in neuroepithelial progenitors of hippocampal neurogenesis. Hippocampus | PubMed
Michael Ratz et al. Cell types and clonal relations in the mouse brain revealed by single-cell and spatial transcriptomics. Nature Neuroscience 2022. Nature Neuroscience | PubMed
Mold, Jeff E, et al. Divergent clonal differentiation trajectories establish CD8+ memory T cell heterogeneity during acute viral infections in humans. PubMed
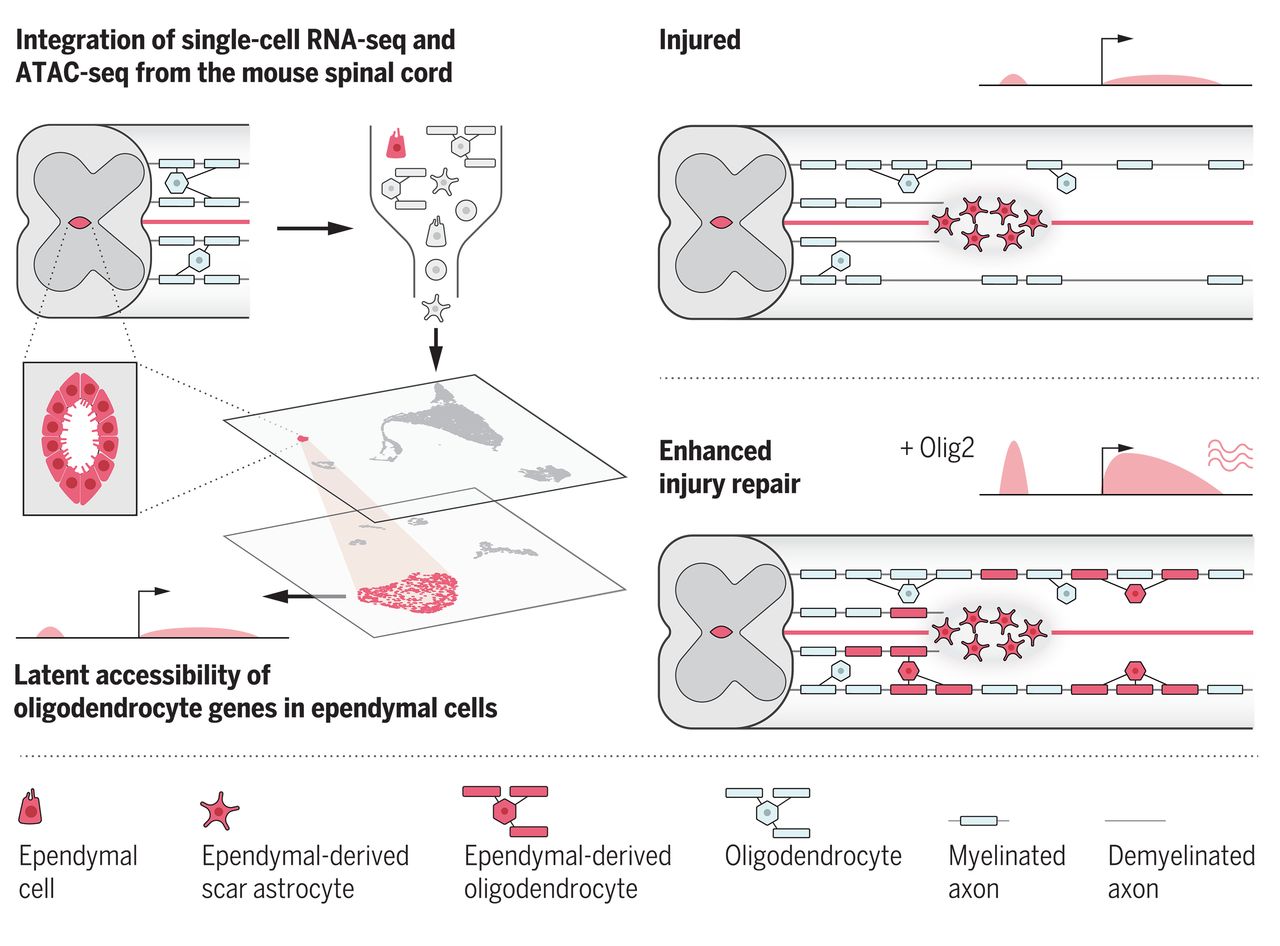
Enric Llorens-Bobadilla, et al. A latent lineage potential in resident neural stem cells enables spinal cord repair. Science, 2020. PubMed | Science
Latent potential in neural stem cells. Through the integration of different layers of genomic information in single cells, we found that the genetic program for oligodendrocyte generation is latently accessible in ependymal neural stem cells of the adult spinal cord. After injury, activating the latent potential by forced OLIG2 expression unfolds efficient oligodendrocyte generation, leading to enhanced myelin repair.
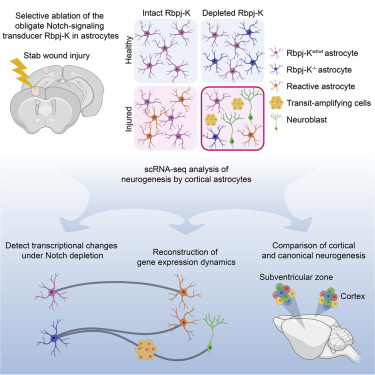
Zamboni, Margherita, et al. A Widespread Neurogenic Potential of Neocortical Astrocytes Is Induced by Injury. Cell Stem Cell, 2020. PubMed
Cortical injury, in combination with Notch signalling depletion, recruits parenchymal astrocytes into a neurogenic program to generate new interneurons. Transcriptional profiling of the astrocytes as they transition towards the neuronal lineage fate, revealed that silencing of Notch initiates a neural stem cell program in both injured and intact cortices, and that neurogenesis occurs after injury independently of reactive gliosis. Furthermore, comparison of transcriptional programs revealed that cortical and subventricular zone neurogenesis develop along similar molecular trajectories.
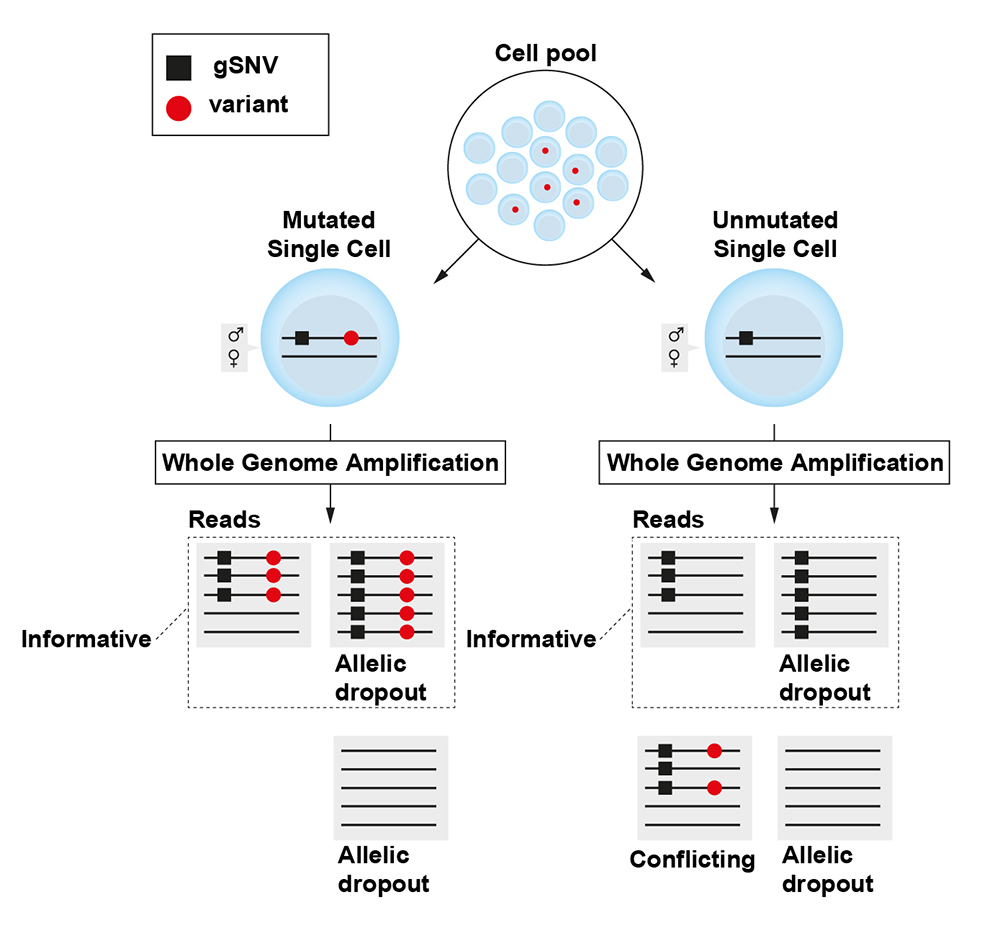
Hård, Joanna; Al Hakim, Ezeddin; Kindblom, Marie, et al. Conbase: a software for unsupervised discovery of clonal somatic mutations in single cells through read phasing. Genome Biology, 2019. PubMed | PDF
Somatic mutations label cells that share a recent common ancestry and can be used to trace cell lineage in humans. Identification of somatic mutations is challenging due to errors and biases in single cell DNA sequencing data. Conbase enables the identification of somatic mutations with high confidence and low false discovery rate by taking advantage of read phasing.
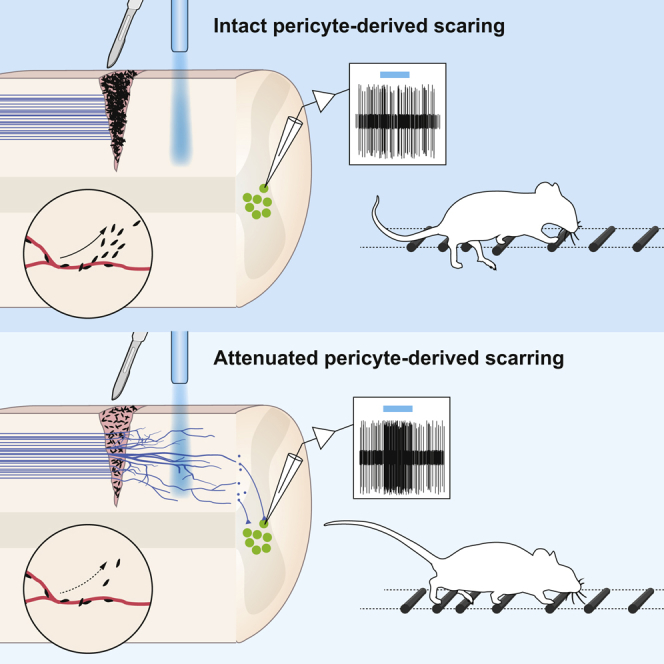
Stenudd, Moa et al. Role of endogenous neural stem cells in spinal cord injury and repair. JAMA Neurology, 2015.
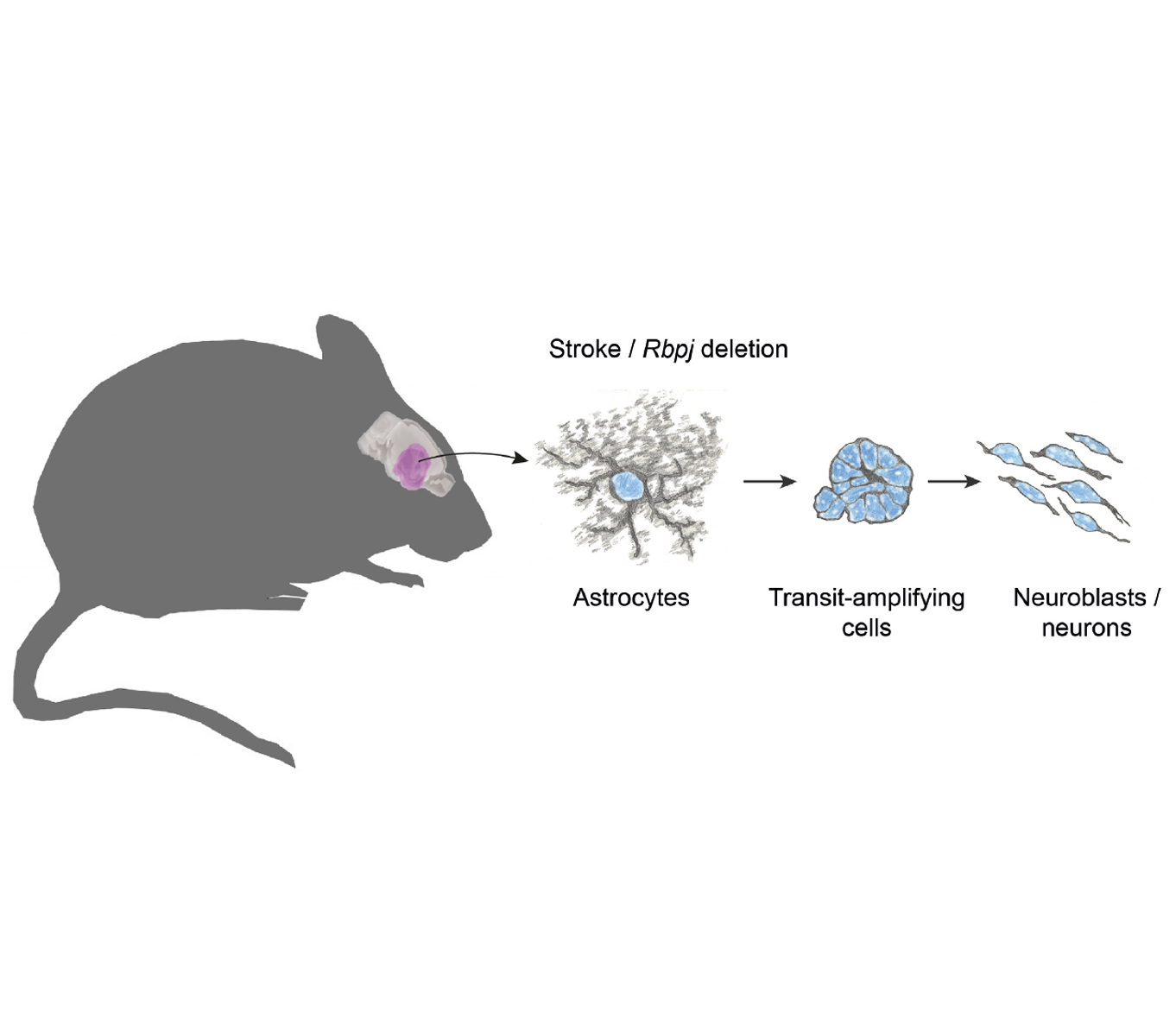
Magnusson, Jens P., et al. A latent neurogenic program in astrocytes regulated by Notch signaling in the mouse. Science, 2014.
Astrocytes in the striatum of adult mice activate a neurogenic program in response to experimental stroke or deletion of the Notch-mediating transcription factor Rbpj. They generate transit-amplifying cells that divide to generate ~40 neuroblasts per starting astrocyte. Some of the neuroblasts mature into neurons.
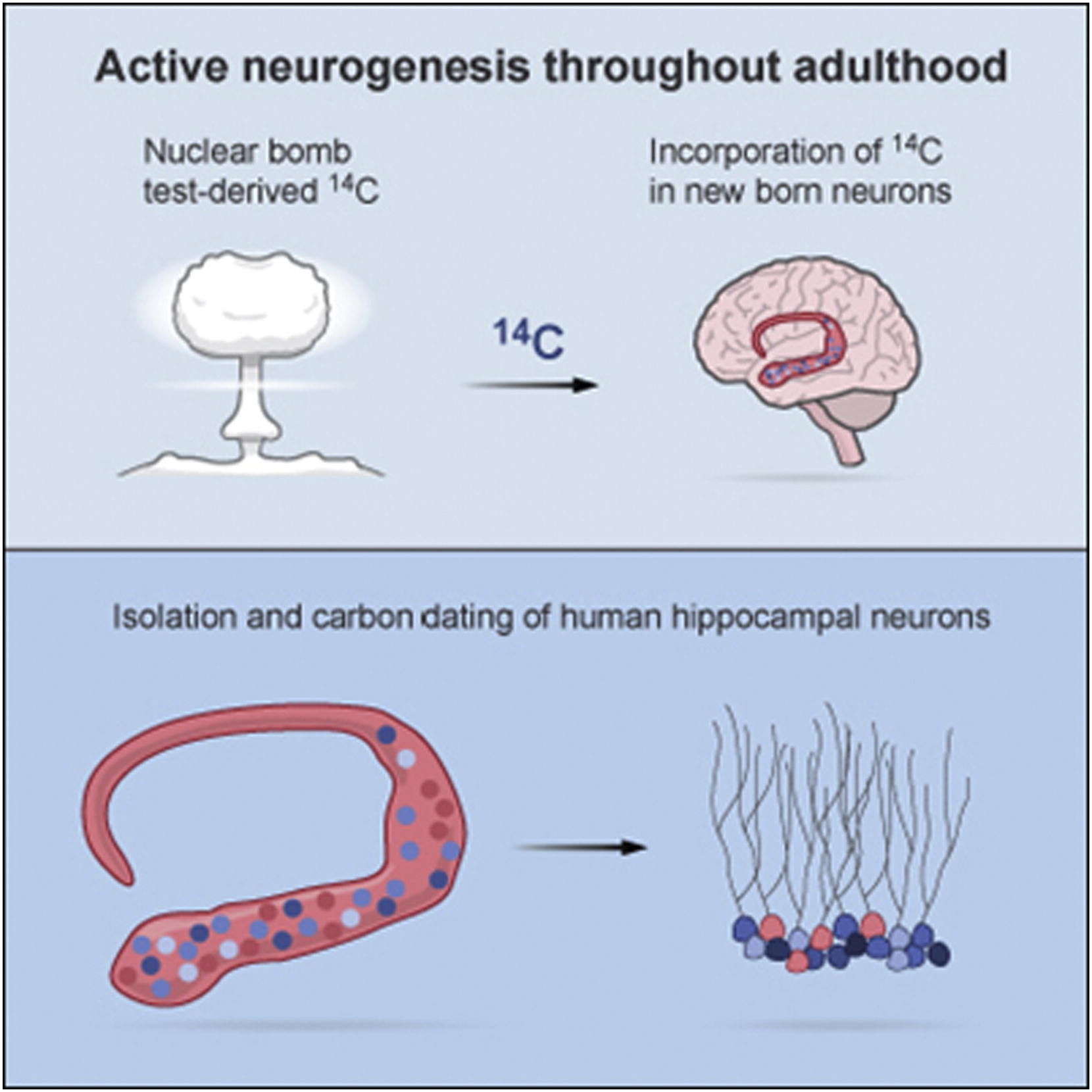
Spalding, Kirsty L.; Bergmann, Olaf, et al. Dynamics of hippocampal neurogenesis in adult humans. Cell, 2013.
Carbon-14 dating of hippocampal neurons in humans showed ongoing neurogenesis in adulthood. A closer look at the dynamics of hippocampal neuron renewal revealed that one-third of all hippocampal neurons may be subject to exchange in an adult human. The annual turnover rate of hippocampal neurons within the group of renewing neurons is 1.75%.